Abstract
Background:JAK mutations are common in myeloproliferative neoplasms (MPN), but are rare in de novo acute myeloid leukemia (AML). Thus, nearly all cases of JAKmut AML can be classified as secondary AML. AML arising from antecedent hematologic malignancies, such as MPNs, is associated with an adverse risk and a high rate of relapse. Little is known about the optimal induction strategy in this subset of disease, either in the upfront setting or at the time of disease relapse. The aim of this study was to evaluate treatment outcomes in JAKmut AML.
Patients & Methods: We analyzed 35 treatment phases of JAKmut AML in 18 patients that evaluated at our institution from January 2016 to December 2021 at VCU Massey Cancer Center. We defined JAKmut AML as AML with JAK1mut, JAK2mut V617F, JAK2mut (NOS), and JAK3mut. Patients were evaluated in both the upfront and relapsed/refractory (R/R) settings. Baseline demographics were obtained, including age, performance status, and comorbidities, as well as disease characteristics, including molecular and cytogenetic profiling, doses of induction regimens, response, and survival. Patients were considered evaluable for response if there was a bone marrow biopsy following a minimum of 28 days of treatment. The event for calculating the overall survival (OS) was the date of death. Patients were otherwise censored at the date of last contact.
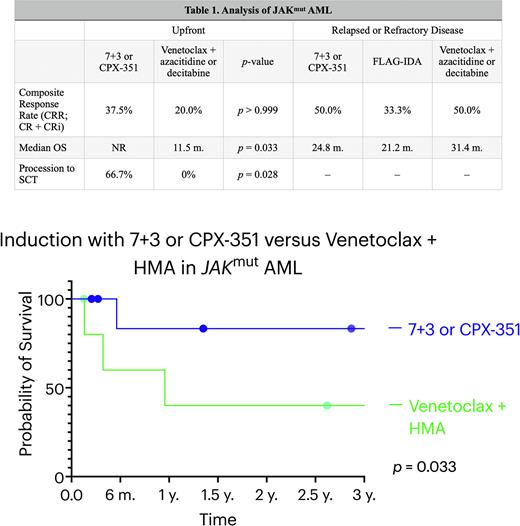
Results: In the upfront setting, 18 patients with JAKmut AML were identified. The median age at diagnosis was 68 years. Of the evaluable patients, four (33.3%) had preceding polycythemia vera, four (33.3%) had primary myelofibrosis, and two (16.6%) had myelodysplastic syndrome. Three (16.7%) patients had ELN 2017 favorable-risk cytogenetics, six (33.3%) were intermediate, and nine (50.0%) were adverse. The most common cooperating mutations were in NRAS (37.5%), ASXL1 (25%), TET2 (25%), NPM1 (18.8%), and NF1 (18.8%). Nine (50.0%) patients underwent induction with an anthracycline and cytarabine (7+3 or CPX-351) and six (33.3%) underwent induction with venetoclax (VEN) and a hypomethylating agent (HMA; decitabine or azacitidine). The composite response rate (CRR; CR + CRi) was 37.5% for anthracycline induction (CR = 2, CRi = 1). In the HMA + VEN cohort, the CRR was 20.0% and the best response was CRi. A total of 66.7% of patients received allogeneic stem cell transplant (SCT) in the anthracycline cohort and no patients received SCT with HMA+VEN (p = 0.028). The median OS was significantly superior in the anthracycline cohort (p = 0.033) and was not reached at a median follow-up time of 34.4 months, while the median OS of the HMA + VEN cohort was 11.5 months. In the R/R cohort of 18 patients, 4 (22.2%) were treated with an anthracycline and cytarabine during re-induction and 4 (22.2%) were treated with FLAG-IDA. The response rate to an anthracycline + cytarabine was 50.0% compared to 33.3% for FLAG-IDA in those evaluable. The median OS of the anthracycline + cytarabine cohort was 24.8 months and 21.2 months in the FLAG-IDA cohort (p = 0.934). Two additional patients were treated with venetoclax + azacitidine or decitabine in the relapsed or refractory setting. One achieved CRi and the other remained on therapy but did not receive a follow-up bone marrow biopsy to evaluate response. The median OS was 31.4 months for venetoclax-based therapy in the relapsed/refractory cohort.
Conclusions: Front-line anthracycline inductions appear superior in JAKmut AML compared to venetoclax-based strategies and were associated with a significantly higher OS. Additionally, patients treated with anthracycline-based inductions were significantly more likely to proceed to SCT. While this may be due to underlying differences in patient selection, it nonetheless highlights the utility of an intensive approach in those that are appropriate candidates. In the relapsed/refractory setting, there were no statistical differences observed between re-induction with 7+3 or CPX-351 and FLAG-IDA, with the median OS appearing similar between both cohorts. Although the venetoclax cohort had two patients, venetoclax-based strategies may be more appropriate for the relapsed/refractory setting.
Disclosures
Maher:BMS: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.